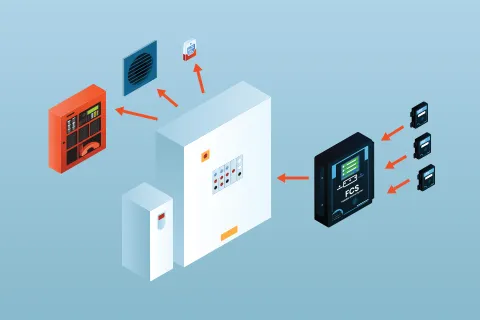
Why Integrate Gas Detection into BMS, BAS or PLC Systems?
Integration with BMS, BAS or other type of control system enables automated ventilation and safety controls and ensures responsive facility management.
Read moreStay informed about the latest developments, product announcements, and industry insights from Critical Environment Technologies.

Integration with BMS, BAS or other type of control system enables automated ventilation and safety controls and ensures responsive facility management.
Read more
While guests are focused on their laps around the pool or personal bests on the treadmill, gas detectors are focused on something else entirely: the air they are breathing.
Read more
Beneath busy cities and inside bus terminals, even small concentrations of CO, NO₂ can accumulate quickly, creating hidden air quality risks that most commuters never see coming.
Read more
When gas detection systems are integrated correctly, they become the intelligence layer that drives ventilation, improving safety, reducing energy use, and extending equipment life.
Read more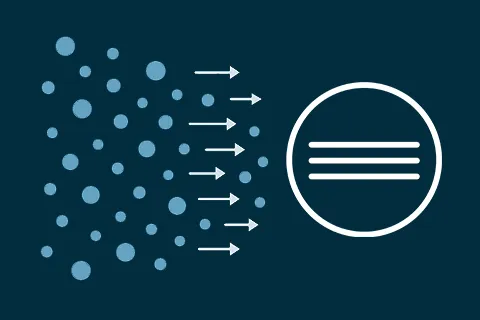
A gas detector is only as good as the gas that reaches it and diffusion is one of the process that decides how well that happens.
Read more
A comprehensive guide to Variable Refrigerant Flow (VRF) HVAC systems and the importance of refrigerant leak detection for building safety.
Read more
Learn to recognize the warning signs of propane gas exposure and what to do if you suspect a leak in your home or facility.
Read more
A fresh year calls for fresh safety checks - make gas detector maintenance your first resolution.
Read more
Improved diffusion model calibration cap for stable calibration across all environments.
Read more
Infrared sensors stand out as a superior solution, offering significant advantages over traditional solid-state sensors.
Read more
Provides continuous monitoring for acute refrigerant leaks in modern energy-efficient buildings utilizing Variable Refrigerant Flow (VRF) HVAC systems.
Read more
One key safety issue that data centers face is the buildup of hydrogen gas in the battery rooms. Hydrogen gas detectors are essential to ensure the safety of people and property.
Read more
With 30+ years of experience, we provide reliable integration. CET gas detection systems seamlessly work with diverse ventilation fans, delivering energy‑efficient solutions that boost safety and cut operating costs.
Read more
A gas detection system in a chiller room is not just a precaution"”it's a critical safety measure.
Read more
Regular maintenance and inspection of the gas detection system helps identify and correct problems before they escalate and lead to false readings or missed detections.
Read more
Regular maintenance and inspection of the gas detection system helps identify and correct problems before they escalate and lead to false readings or missed detections.
Read more
The sensor coverage range for CO and NO2 gas detectors in parking garages is larger than many other applications. Find out why.
Read more
Infrared refrigerant gas sensors offer a powerful combination of accuracy, reliability, and low maintenance, making them an excellent choice for modern HVAC and refrigeration systems.
Read more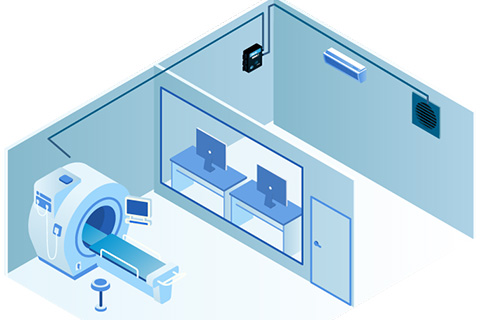
Designed to replace the legacy DCC-SD and DCC-MRI systems, the cGas-SD brings a new level of performance, usability, and flexibility to gas detection in hard-to-access environments.
Read more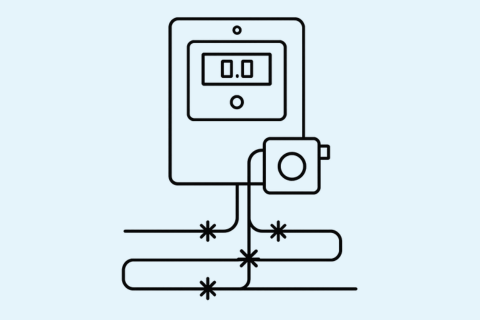
While multi-point sample draw systems can seem efficient, they come with significant risks and drawbacks that may outweigh their benefits in critical gas detection applications.
Read more
Oxygen is essential for life, yet it can also pose significant risks. Ensuring that oxygen levels are properly monitored and maintained is vital for safeguarding human health and safety.
Read more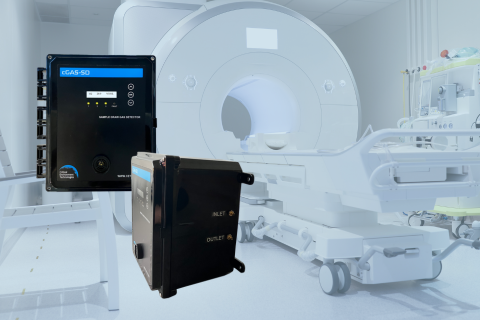
The cGas-SD Sample Draw System elevates your safety standards with precision monitoring.
Read more
Gas detectors are vital safety devices designed to monitor and measure potentially lethal gases in various environments, safeguarding lives, property, and the environment.
Read more
The height at which to mount a fixed gas detector depends on the density of the target gas relative to air and can also depend on the on the type of application.
Read more
The integration of gas detection systems in greenhouse production and oil extraction facilities is essential for maintaining safety and efficiency.
Read more
At CET, we've been at the forefront of the gas detection evolution. Our journey over the past 30 years has been marked by continuous improvement and a commitment to excellence.
Read more
Ammonia is a corrosive, toxic gas or liquid used in manufacturing and refrigeration. Exposure to high levels of ammonia can be fatal. Learn the ins and outs about ammonia gas detection systems.
Read more
A2L refrigerants offer a safer and more environmentally friendly alternative to traditional refrigerants, but they require careful handling and specific safety standards to mitigate potential hazards.
Read more
With practice and the right tools, this conversion becomes simple—an essential part of accurate, compliant gas detection.
Read more
By incorporating a low temperature package, gas detectors can provide reliable performance and safety across a wider range of environments.
Read more
Ensuring safety with gas detectors involves proper usage, calibration, and maintenance to effectively monitor hazardous gases and protect lives and property.
Read more
This technical resource from Critical Environment Technologies provides essential information for gas detection professionals, facility managers, and safety engineers.
Read more
Bedbug infestation is an extremely common problem worldwide.
Read more
When choosing between fixed and portable gas detectors there are many factors to consider to ensure the proper monitoring measures are being taken to ensure the health and safety of occupants.
Read more
When considering the hazardous gases in a brewery, CO2 is likely the first to come to mind. While it plays a crucial role in brewing, other gases like ammonia, nitrogen, and ozone can also be present.
Read more
Gas detectors rely on accurate signals to detect gas concentrations. Proper wiring prevents interference, signal loss, or noise that could affect the detector's performance.
Read more
Both NO and NO2 are produced during the combustion process and are emitted into the air through the exhaust of gasoline run vehicles. What other similarities and differences do the two gases have?
Read more
Wildfire smoke contains fine particles that can irritate the respiratory system, damage lung tissue, and increase long‑term health risks. Even indoors, you’re not fully protected from its harmful effects.
Read more
They are all around you, be aware of them and stay safe.
Read more
The cGas‑SC Self‑Contained Controller—offering versatile customization and reliable gas detection performance.
Read more
Many gases—some detectable by humans, others not—can leak into the spaces where we work and play, becoming dangerous when they exceed safe concentrations.
Read more
Solid‑state sensors are versatile and can detect many gases, but they require clean, stable environments with minimal temperature and humidity fluctuations.
Read more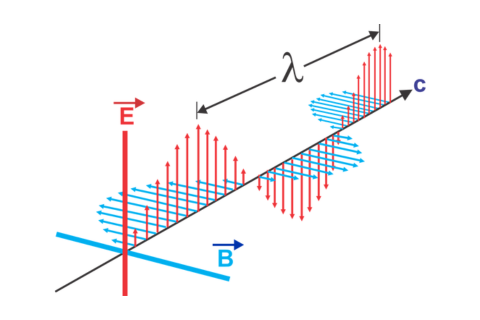
Non-dispersive infrared sensors accurately detect CO2 and refrigerant gases without cross interference from toxic or combustible gases because the absorption of the infrared light occurs at a wavel...
Read more
The potential for exposure to hazardous gases should be a concern in many workplace environments.
Read more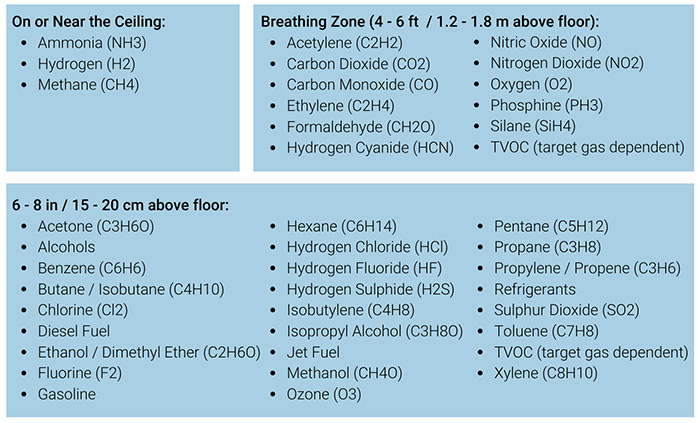
Proper sensor placement is critical for effective gas detection. The mounting height and location of sensors directly impacts their ability to detect hazardous conditions and protect personnel. Thi...
Read more
Catalytic sensors are used for detecting combustible, or flammable gases, such as methane, propane and hydrogen. They are a safe and reliable combustible sensor technology that has been used for man..
Read more
Electrochemical sensors are effective and offer very good performance for monitoring toxic gases and oxygen. Their lifespan depends on several factors.
Read more
Gas detection transmitters play a crucial role in safeguarding lives and property by monitoring and detecting hazardous gases in the environment. These devices consist of various components, with sens
Read more
Need product NOW?! Check out our most popular products that are now available for quick ship, in as little as 2 days!
Read more
CET offers CO2 gas detectors for indoor air quality and for life, health and safety applications with various ranges, units of measure and alarm levels that are fully programmable in the field.
Read more
Many recreational facilities offer both skating and swimming amenities, two very different environments with several different potential gas hazards. How many gas detectors are really needed?
Read more
Formaldehyde is a chemical with no colour and an unpleasant smell. At room temperature it is a gas and it also comes in a liquid solution form. Formaldehyde can cause irritation of the skin, eyes, no.
Read more
CO2 is naturally occurring all around us and in low concentrations it is not the least harmful. However, CO2 has the potential to exist in unhealthy concentrations in a multitude of applications which
Read more
Boiler rooms play a vital role in building operations, but they also pose serious safety risks due to the potential buildup of hazardous gases. Typically located in unfrequented areas, continuous gas detection monitoring is crucial to ensure personnel remain aware of any dangerous conditions.
Read more
Advancements in gas detection technology has made energy and cost management possible by connecting the two systems together and putting the gas detection system in the driver's seat to run the ventil
Read more
Calibration frequency is one of the most commonly asked questions concerning the use of gas detection instruments. The answer depends on a number of factors, including the type of gas detection system
Read more
Everyday exposure to indoor pollutants can cause you to lose life expectancy and you may die years earlier then you should!
Read more
Most malfunctions are discovered at the time of calibration or bump testing, meaning at some point between calibrations the gas monitor manifested a problem.
Read more
Gas detectors play a crucial role in building automation and control systems, providing real-time data on a range of factors that impact the safety and comfort of building occupants.
Read more
Navigating gas detection regulations can be complex. CET makes it simple with our Sense Series of compliance solutions: ChillSense, BoilerSense and ParkSense each tailored to specific applications
Read more
Remote audible/visual devices offer superior notification because they are loud and bright and more likely to promote a generalized and appropriate human response to an emergency situation.
Read more
CET has launched a brand new non-dispersive infrared, dual beam gas detector for accurate, low level detection of a wide variety of refrigerants, including A2L refrigerants, SF6 and CO2 (R744).
Read more
Today's buildings, whether it is a home, office, manufacturing plant, hospital, etc., are more energy efficient; however, they also foster a potentially harmful environment. Indoor Air Quality (IAQ) h
Read more
CET"s high range A2L refrigerant gas detectors satisfy the ASHRAE 15-2024 standards, complying with the 25% LFL alarm point and ensuring comprehensive safety and compliance for all your commercial ref
Read more
It only takes a spark to start a fire or cause an explosion, especially when flammable gases or vapours are present under the right conditions.
Read more
Natural gas is a flammable gas that is commonly used in building heating systems such as schools, universities and dormitories. It consists primarily of methane gas. The source of the natural gas is..
Read more
Gas cooking equipment such as grills, stoves and fryers in restaurants, hotels and other commercial kitchens often use natural gas or propane for an energy supply. If a burner was left on or a fitting
Read moreThis technical resource from Critical Environment Technologies provides essential information for gas detection professionals, facility managers, and safety engineers.
Read more
A gas detection system is an important and required component for ensuring the health and safety of everyone in an aquatic recreation facility. Below is some general information about both gases
Read more
Classification Toxic, oxidizing agent, powerful disinfectant Molecular Formula O3 Molecular Weight 47.997 g/mol Fire Hazard Rating Risk of fire and explosion on contact with combustible substances M..
Read more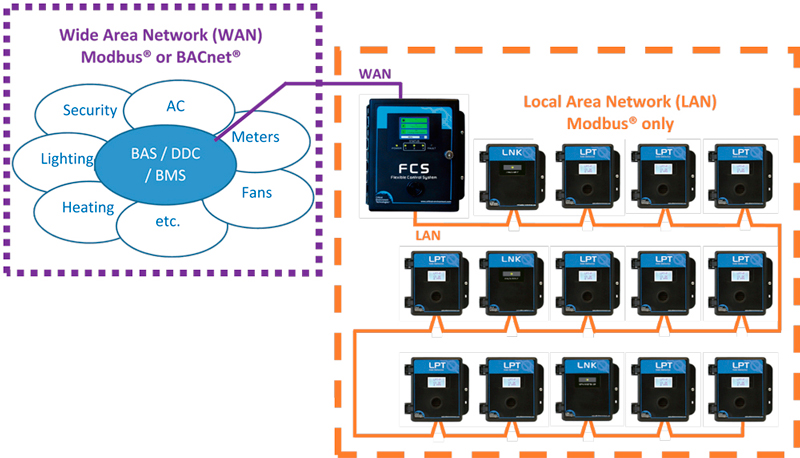
Modern gas detection systems support various communication protocols for integration with building automation and control systems. Understanding these options helps optimize system design.
Read more
Bump testing a gas sensor involves flowing a concentration of gas that is slightly higher than the Low alarm setpoint of the gas detector for a short period of time (1-2 minutes or less).
Read more
Classification Inorganic, toxic gas Molecular Formula NH3 or H3N Molecular Weight 17.031 g/mol Fire Hazard Rating Slight to moderate, on its own The presence of oil or other combustible materials incr
Read more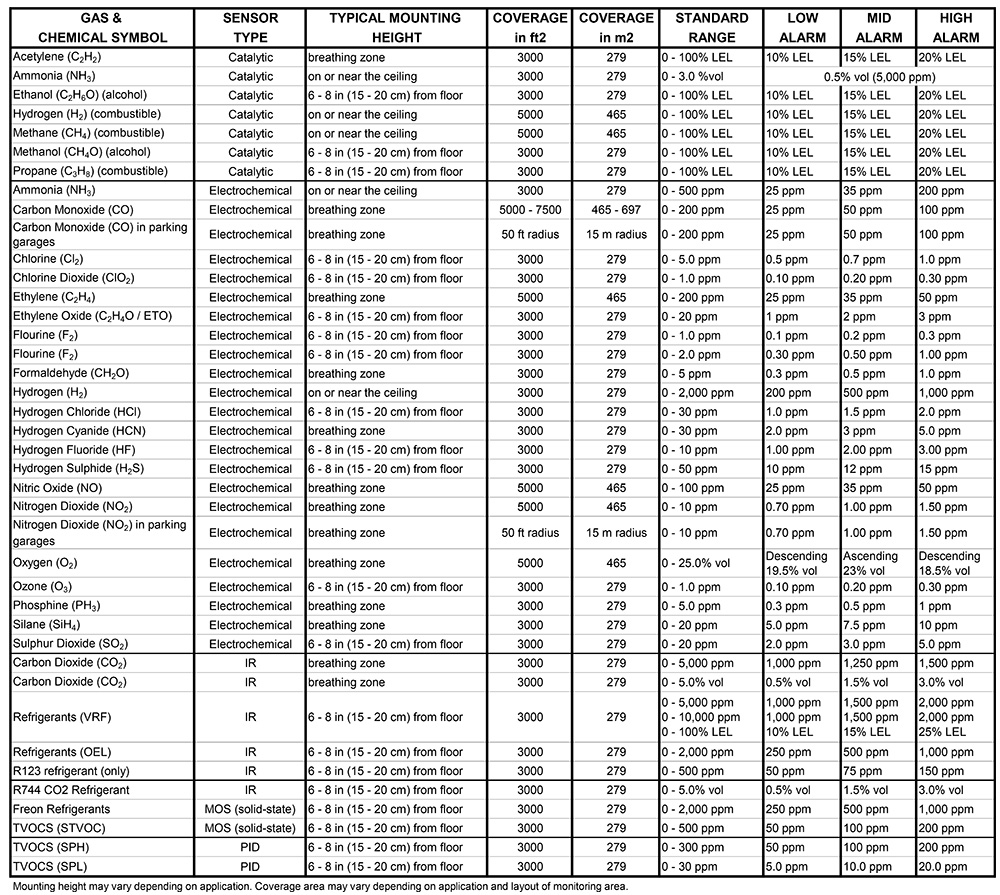
Properly configured alarm setpoints are crucial for early warning while avoiding nuisance alarms. The following guidelines are based on occupational exposure limits and industry best practices for ...
Read more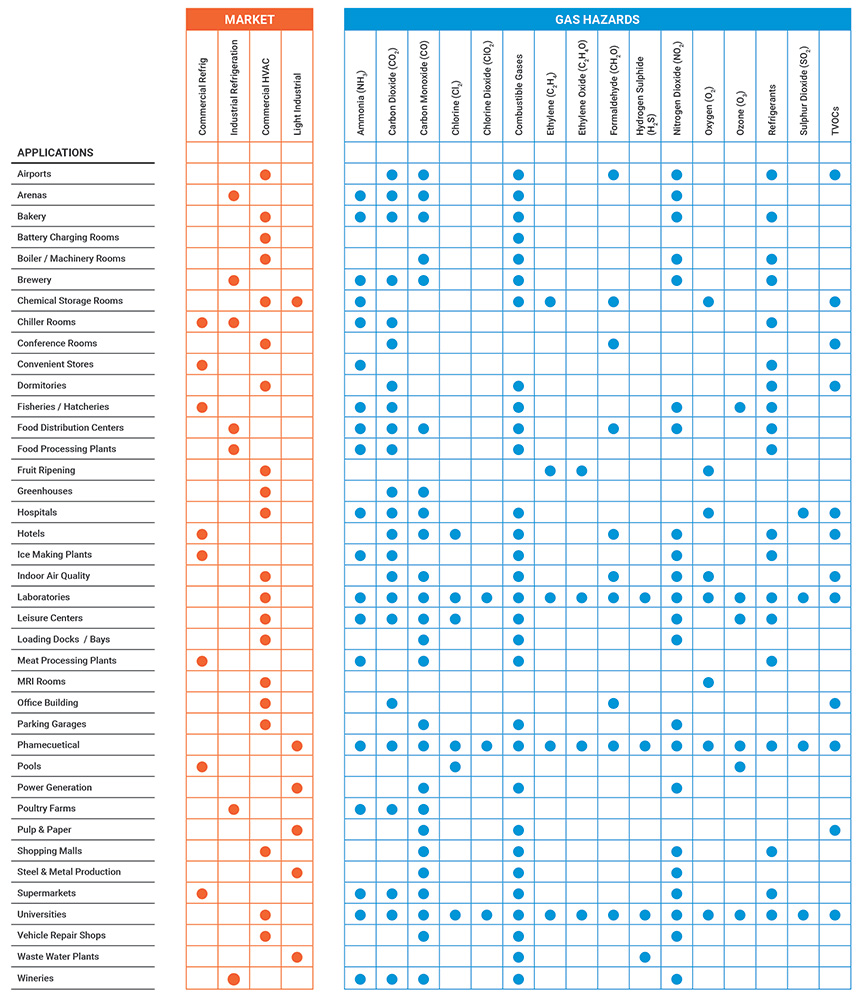
Different industries and applications face different gas hazards. This reference chart helps identify the relevant gases for common market segments.
Read more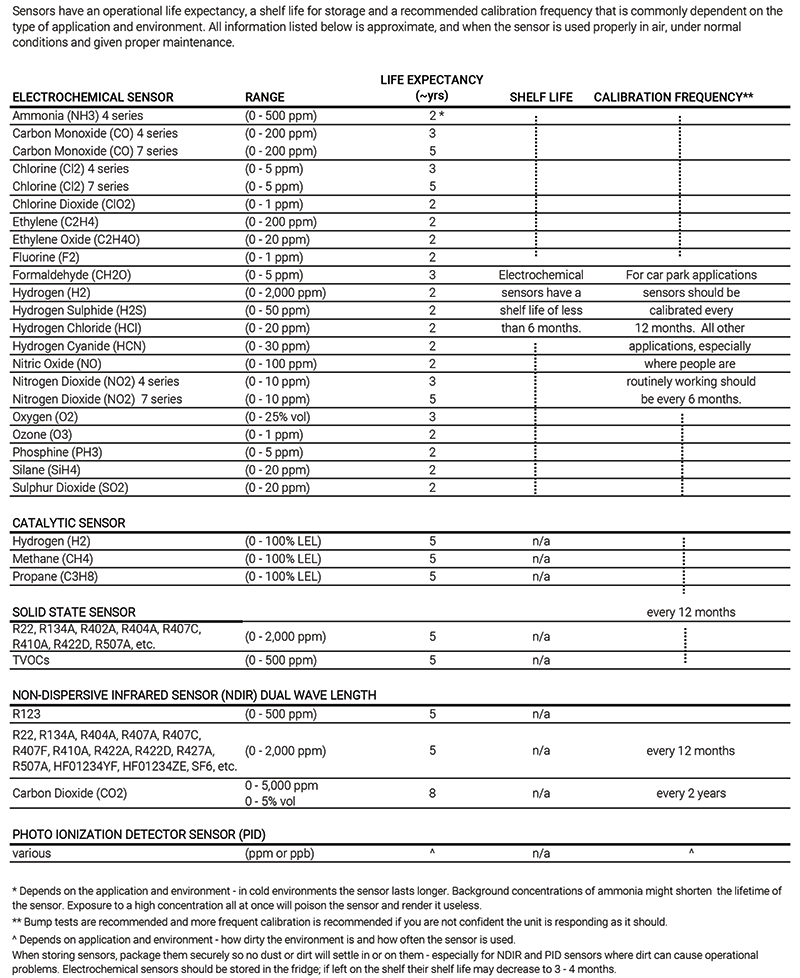
Understanding sensor lifespan is essential for planning maintenance schedules and ensuring continuous protection. Different sensor technologies have varying life expectancies based on their operati...
Read moreThe gas detection industry uses many specialized abbreviations and acronyms. This reference guide covers the most commonly encountered terms.
Read moreConverting between different units of gas concentration is a common requirement in gas detection. This reference guide provides the key conversion factors used in the industry.
Read moreProper sensor placement is critical for effective gas detection. The mounting height and location of sensors directly impacts their ability to detect hazardous conditions and protect personnel. Thi...
Read moreThis technical resource from Critical Environment Technologies provides essential information for gas detection professionals, facility managers, and safety engineers.
Read moreElectrochemical sensors are the most common technology for detecting toxic gases at low concentrations. These sensors offer excellent sensitivity, selectivity, and response times for workplace safe...
Read moreIndoor air quality monitoring is increasingly important for occupant health and comfort. This Q&A addresses common questions about IAQ sensing and control.
Read moreIndoor air quality monitoring is increasingly important for occupant health and comfort. This Q&A addresses common questions about IAQ sensing and control.
Read more
Carbon monoxide (CO) and carbon dioxide (CO₂) are often confused, but they are fundamentally different gases with distinct properties, hazards and gas detection requirements. Understanding these differences is essential for proper gas detection system design.
Read more
The FCS offers an incredibly flexible control structure using alarm level settings, time of day settings, advanced logic, multiple priority levels per zone, etc. meeting the requirements of Californi.
Read more
With the legalization of marijuana in many North American regions, the cannabis cultivation industry is booming.
Read more
Critical Environment Technologies is pleased to announce the release of our FCS Flexible Control System multichannel controller.
Read more
Regular testing and calibration are essential to ensure gas detection equipment functions correctly when needed. This guide covers the key procedures for verifying detector operation.
Read more
Monitoring ethylene and carbon dioxide in ripening rooms allows for more efficient control and optimization of the ripening process, a higher safety level and better production.
Read more
Choosing the right enclosure rating can make or break the reliability of electrical equipment in different types of environments. Two systems dominate the conversation—IP ratings and NEMA ratings—and while they serve a similar purpose, they measure protection in different ways.
Read more
Strategic and reliable gas detection solutions for wastewater treatment plants.
Read more
Depending on the number and placement of gas detectors in your facility, you might be looking at the task of calibrating them as never-ending. Should they all be calibrated at the same time?
Read more
Important differences between types of gas detectors make using residential models in commercial or industrial environments insufficient, dangerous and unlawful.
Read more
Gas cylinders are made by many different manufacturers, are available in a variety of sizes, can be disposable or refillable and filled with a low, high or pressurized concentration.
Read more
It only takes a spark to start a fire or cause an explosion, especially when flammable gases or vapours are present under the right conditions.
Read more
While both residential and commercial gas detectors serve safety purposes, they are designed for very different applications and regulatory requirements. Using the wrong type can create serious saf...
Read more
Calibration gas cylinders have limited shelf lives that vary based on the gas mixture and concentration. Proper cylinder management ensures accurate calibration results.
Read more
Many gas hazards are present in various locations within community recreational facilities and should be monitored by multiple gas detectors.
Read more
In the past, vehicle exhaust gas detectors have been installed on or near the ceiling. In contrary, Critical Environment Technologies Canada Inc. (CETCI) has always recommended that they be installed.
Read more
There are many health risks associated with the beauty industry due to the chemicals found in the products. We are all affected by these chemicals every time we go to the beauty salon. Many of the pro
Read more
Carbon dioxide, also known as CO 2 , is a very well known gas type but not everyone knows how deadly it is. Most people know it as a gas that humans exhale but commercially, it is manufactured and shi
Read more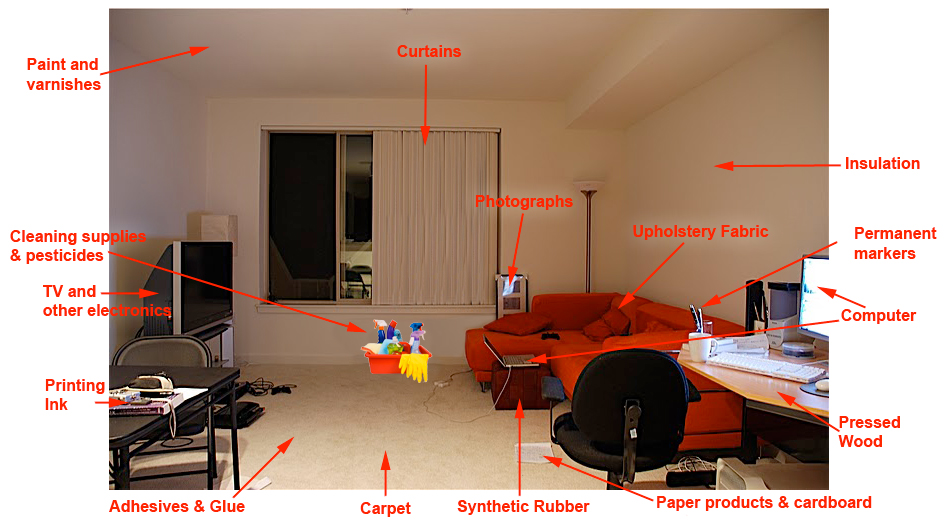
Chemicals that emit gas are called volatile organic compounds (VOC).
Read more